The program includes:
- Initial presentation in the clinic
- history taking
- general clinical examination
- laboratory tests:
- complete blood count
- biochemical analysis of blood
- urinalysis
- TSH-basal, fT3, fT4
- PSA blood test
- tests for urogenital infections
- indicators of inflammation
- indicators of blood coagulation
- ultrasound scan of the urogenital system
- renal scintigraphy
- PSMA PET-CT (on indication, additional cost)
- PSMA treatment
- full body scintigraphy after 24 hours
- full body scintigraphy after 48 hours
- symptomatic treatment
- cost of essential medicines
- nursing services
- elaboration of further recommendations
- stay in the hospital with full board
- accommodation in a room with 2 beds
How program is carried out
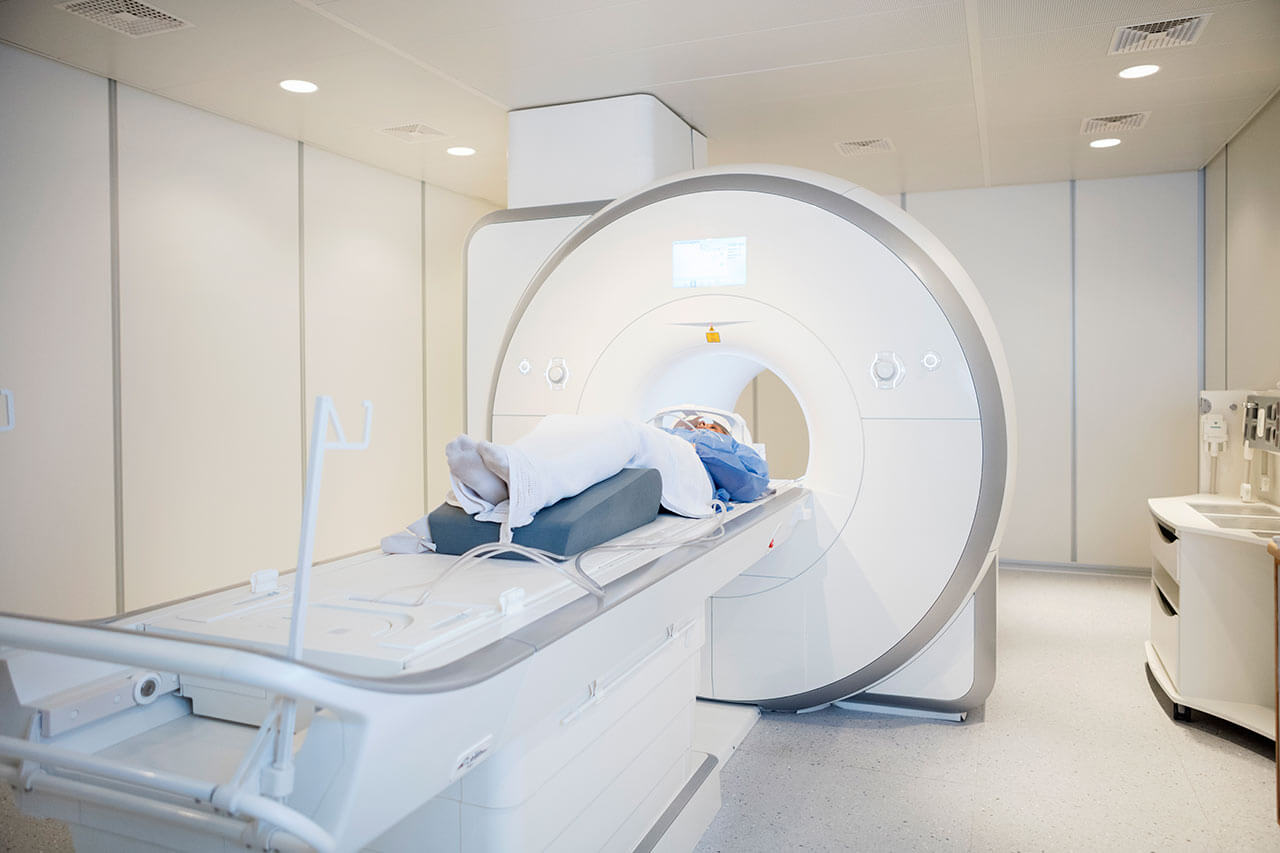
During the first visit, the doctor will carry out a general physical examination and go through the results of your previous laboratory and instrumental tests. After that, you will undergo any necessary additional tests, such as an assessment of your liver and kidney function, a scintigraphy of your skeleton and salivary glands, a PSMA PET/CT (additional cost). This will allow the doctor to calculate your individual dosage of the radionuclide.
PSMA therapy with Lutetium-177 is carried out via intravenous administration of a solution containing the radioactive isotope, Lutetium-177. The solution is injected through a catheter. This is a short procedure, as the infusion usually takes no more than 20 minutes.
During the procedure, you will need to apply cooling bags to your salivary glands, as Lutetium-177 partially accumulates in the salivary glands, affecting their function and causing a dry mouth. You will also receive intravenous saline solutions to protect your kidneys.
After the infusion of Lutetium-177, you will stay in a specially equipped (radiation-shielded) ward for 48 hours. The drug is quickly excreted by the kidneys, and after 48 hours you will no longer be dangerous to others. During these 48 hours, you can read, use a mobile phone, tablet or computer – none of these devices will be a source of radiation in the future.
Follow-up examinations include whole-body scintigraphy and computed tomography in 24 hours, and then again 48 hours after the procedure.
Required documents
- Medical records
- MRI/CT scan (not older than 3 months)
- Biopsy results (if available)
Service
You may also book:
 BookingHealth Price from:
BookingHealth Price from:
About the department
The Department of Nuclear Medicine at the University Hospital of the Ludwig Maximilian University of Munich offers the full range of modern methods of radioisotope-based diagnostics and treatment. The department provides outpatient and inpatient medical care. In the field of diagnostics, special attention is paid to positron emission tomography (PET), including in combination with computed tomography (PET/CT), scintigraphy, single photon emission computed tomography (SPECT), and SPECT/CT. These examinations are most often performed for suspected cancers, neurological disorders, and heart disease. Radioisotope-based therapies can be used to treat benign and malignant thyroid diseases, benign joint lesions, prostate cancer, neuroendocrine tumors, primary and secondary liver tumors, and bone metastases. The department is the largest facility of this kind in Germany and is deservedly proud of its prestigious international ISO 9001:2015 certification. The Head Physician of the department is Prof. Dr. med. Matthias Brendel.
An integral part of the department's clinical practice is high-precision diagnostic procedures such as scintigraphy, PET, PET/CT, SPECT, and SPECT/CT scans. The above-mentioned tests are of particular value in diagnosing patients with suspected malignant diseases of various localizations, assessing the state of the sentinel lymph nodes, brain, and spinal cord, as well as assessing the condition of the heart vessels and diagnosing a heart attack. Modern radioisotopes allow doctors not only to see the anatomical structure of the organ but also to assess its function, blood flow, and metabolic processes. Nuclear diagnostic methods often make it possible to detect pathology at an early stage when a patient does not experience any symptoms, which is not always possible with the help of laboratory and hardware diagnostic methods.
Since August 2013, the department has had a new center for the production of radiopharmaceuticals. The center is equipped with a cyclotron, which allows for the production of radioactive drugs based on the GMP standard in compliance with the current requirements for radiation protection and prescribing medicines. Thanks to this, the department receives the necessary high-quality and safe substances for diagnostics and radiopharmaceuticals for customized therapy in a shorter time. The doctors can thus provide patients with top-class treatment.
The team of the department's doctors also has perfect professional skills and exceptional experience in providing treatment with radiopharmaceuticals. Radioiodine therapy for benign and malignant thyroid diseases, pain management for skeletal metastases, radiosynoviorthesis for joint lesions, Lu-177-DOTATATE therapy for neuroendocrine tumors, Lu-177-PSMA therapy for prostate cancer, and selective internal radiation therapy (SIRT). These treatment programs are often elaborated in collaboration with doctors from other medical specialties (oncologists, endocrinologists, gynecologists, surgeons, orthopedists), for example, at the Comprehensive Cancer Center (CCC) or the Breast Center.
There are 16 beds available to accommodate inpatients in the department. The wards in the medical facility are different from those that patients are used to seeing in conventional clinics, since it is important for doctors to meet strict radiation protection rules. The duration of a hospital stay for radionuclide therapy is usually about one week. At the same time, the mandatory stay in a special room with radiation protection systems is 2-3 days. The indisputable advantages of radionuclide therapy are its high efficiency, painlessness, and absence of side effects.
The department's range of medical services includes:
- Diagnostic options
- Scintigraphy
- Detection of the source of bleeding
- Scintigraphy to detect inflammatory processes
- Brain scintigraphy
- Testicular scintigraphy
- Bone marrow scintigraphy
- Liver scintigraphy
- Positron emission tomography (PET)
- PET/CT for suspected cancer
- PET for suspected neurological/neurooncological pathologies
- PET and PET/CT for other diagnostic tasks (for example, in cardiology, etc.)
- SPECT and SPECT/CT
- Scintigraphy
- Therapeutic options
- Radioiodine therapy for benign and malignant thyroid diseases
- Lu-177-PSMA-DKFZ-617 therapy for prostate cancer
- Xofigo® (Ra-223) therapy for bone metastases
- Peptide receptor radionuclide therapy for neuroendocrine tumors: Lu-177-DOTATATE therapy and 90Y-DOTATATE therapy
- Metaiodobenzylguanidine treatment (MIBG therapy) for pheochromocytomas and other tumors
- Selective internal radiation therapy (SIRT) for primary and metastatic liver tumors
- Radiosynoviorthesis (RSO) for pathological joint lesions
- Other diagnostic and treatment methods
Curriculum vitae
Higher Education and Professional Career
- Since January 2024 Head Physician, Department of Nuclear Medicine, University Hospital of Ludwig Maximilian University of Munich.
- Since April 2023 W2 Professor for Translational Molecular Imaging and Senior Physician at the Department of Nuclear Medicine, University Hospital of Ludwig Maximilian University of Munich.
- 2018 - 2023 Acting Head Physician, Department of Nuclear Medicine, University Hospital of Ludwig Maximilian University of Munich.
- Medical studies, Ludwig Maximilian University of Munich.
Photo of the doctor: (c) LMU Klinikum
About hospital
According to the Focus magazine, the University Hospital of Ludwig Maximilian University of Munich is regularly ranked among the best medical institutions in Germany!
The hospital is the largest multidisciplinary medical facility, as well as a leading research and training center in Germany and Europe. The hospital is proud of its bicentenary history and tirelessly confirms its primacy at the national and international levels. The outstanding quality of medical care is complemented by highly productive research activities, thanks to which many effective diagnostic and therapeutic methods, saving people’s lives, have been presented in medical practice.
The medical facility includes two main buildings, Grosshadern and Innenstadt. The hospital has 29 specialized departments, 53 interdisciplinary centers, 11 institutes, and many sections. More than 500,000 patients are treated here every year, which indicates the hospital's excellent reputation. A large and highly professional medical team, consisting of 1,800 doctors and 3,300 nursing staff, works for the benefit of patients. The hospital has 2,000 beds to accommodate patients.
The hospital's infrastructure deserves special attention: advanced diagnostic equipment that allows doctors to detect the slightest pathological changes in the human body, the latest operating rooms with highly efficient monitoring systems, robot-assisted surgical systems that facilitate sparing operations, and proper postoperative care.
Excellent technical resources and highly professional medical staff are undoubtedly the hospital's pride, but the medical facility also pays attention to the patient's comfort and to a humane attitude toward their life situation. When providing the necessary medical care, doctors and nursing staff always show a friendly attitude, inform patients in detail about the upcoming diagnostic and therapeutic procedures, gladly answer all questions of interest to patients, and provide moral support during the therapeutic process.
The hospital has many prestigious quality certificates, including a DIN EN ISO 9001 certificate, an IQM certificate, an endoCert certificate, certificates from the German Cancer Society (DKG) for treating various types of cancer, the German Cardiac Society (DGK), the German Society for Orthopedics and Trauma Surgery (DGOU), etc. Thus, patients can count on the best possible treatment outcome due to the use of the most effective and, at the same time, sparing therapeutic techniques.
Photo: (с) depositphotos
Accommodation in hospital
Patients rooms
The patients of the University Hospital of Ludwig Maximilian University of Munich live in comfortable, spacious, single and double patient rooms with a modern design. Each room is equipped with an ensuite bathroom with a shower and toilet. The furnishing of a standard patient room includes a comfortable bed, the position of which can be adjusted using the remote control, a locker for storing personal belongings, a TV, and a telephone. Also, if desired, you can connect to the Internet. In addition, patients can opt for enhanced-comfort rooms, with a safe, a fridge, and upholstered furniture.
The hospital has an excellent infrastructure. The medical facility’s area houses a bank, ATMs, a hairdresser, shops with a wide range of food, drinks, newspapers, magazines, and personal hygiene items, play areas for children, and a beautiful garden for walking, etc.
Meals and Menus
The patient and his accompanying person are offered a daily choice of three menus, including a vegetarian one. If you are on a specific diet for any reason, you will be offered an individual menu. Please inform the medical staff about your dietary preferences prior to the treatment.
Further details
Standard rooms include:
Religion
Religious services are available upon request.
Accompanying person
Your accompanying person may stay with you in your room or at a hotel of your choice during the fixed program.
Hotel
You may stay at a hotel of your choice during an outpatient program. Our managers will help you to choose the best option.
The hospital offers a full range of laboratory tests (general, hormonal, tests for infections, antibodies, tumor markers, etc.), genetic tests, various modifications of ultrasound scans, CT scans, MRI and PET/CT, angiography, myelography, biopsies, and other examinations. Treatment with medications, endoscopic and robotic operations, and stereotaxic interventions are carried out here, modern types of radiation therapy are also used. The hospital offers patients all the necessary therapeutic techniques.
- Allogeneic bone marrow transplantation
- Microsurgical transplantation of head and neck tissues
- Microsurgical resection of brain tumors with intraoperative fluorescence
- Minimally invasive treatment of spine pathologies
- Joint replacement with postoperative rehabilitation (fast track program)
Patients with benign and malignant neoplasms of various localizations, pathologies of arteries and veins, herniated discs, osteoporosis, congenital and acquired pathologies of the musculoskeletal system, benign and malignant pathologies of the mammary gland, and other pathologies.
Which specialties of the University Hospital of Ludwig Maximilian University of Munich are the best?
- Interventional and diagnostic neuroradiology
- Vascular surgery
- Cardiac surgery
- Mammalogy
- Gastroenterology and hepatology
Over 1,700 highly qualified doctors work at the hospital.