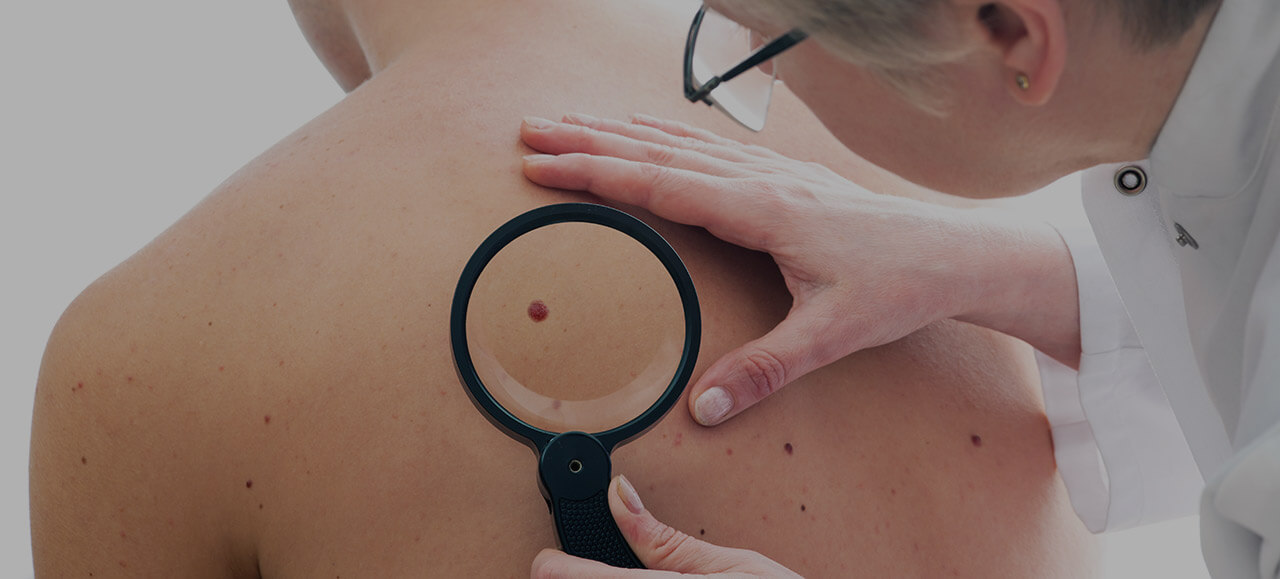
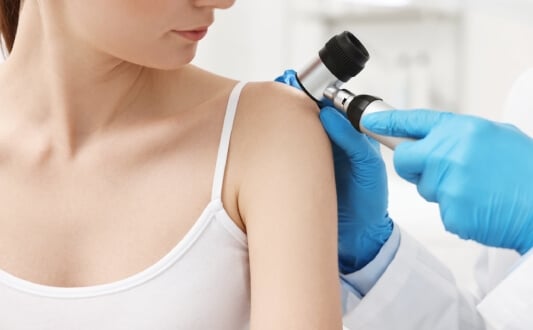
Malignant melanoma is highly aggressive form of skin cancer. For many patients with malign melanoma in an advanced stage, there was little hope until recently. In recent studies, there have been new and hopeful developments in the medical world.
Making breakthroughs for melanoma patients are two newly licensed drugs that show advantages for survival rate.
Each year about 19,000 people in Germany are confronted with that diagnose. Most of them are in an early stage of the disease, luckily for them the tumor can be removed surgically. For other patients, it is too late to remove the tumour since it has spread to lymph nodes, lung, liver, brain or bone.
Patients with distant metastasis have a bad prognosis, without treatment their life expectancy is about 6 to 9 months only. The drugs that were used were not effective. Most times they could only decrease the size of the tumors and reduce symptoms. Advantages for the survival couldn’t be proved without doubt. In the light of this desolate situation the results of the clinical studies with Ipilimumab and Vemurafenib had to make an impact.
Content
- Survival advantages compared to vaccines or Dacarbazin
- Loosen the immune break or interrupt signal paths
- No light without shadow
Survival advantages compared to vaccines or Dacarbazin
Both drugs proved to be superior to the comparing therapies in clinical studies. The median survival under Ipilimumab was 10 month, under the tumor vaccine GP100 on the other hand only 6 months. Further evaluation showed, that 29% of the patients responded to the therapy with Ipilimumab (GP 100 11%), and that after one and two years, 46% respectively 24% of the patients were still alive (GP100: 24% respectively 14%). A few patients of the Ipilimumab group were alive even after 4 years.
Vemurafenib was tested in comparison to the chemotherapeutic Dacarbazin. An intermediate evaluation showed, that after 6 month treatment with Vemurafenib, 84% of the patients were still alive, with Dacarbazin only 64%. 48% of the patients responded to Vemurafenib, to Dacarbazin close to 6%. The median survival time under Vemurafenib was 13 months while Dacarbazin was under 10 months.
To the frustration of doctors and patients, those results must have been like a silver lining. After the results were posted, eager patients flooded the forum discussions with their interest in this new drug treatment. The companies responded to the demand by installing programs for the Compassionate Use or Expanded Access to the drugs. That means the use of not yet licensed drugs on patients, which are not included in clinical studies. Meanwhile, both drugs have been lice the EU, Ipilimumab since July 2011, Vemurafenib since February 2012. What doctors appreciate especially is that their new weapons against the malign melanoma are obviously not only more effective than other drugs, they also have completely different mechanisms of action.
Loosen the immune break or interrupt signal paths
Ipilimumab unfolds its effect by loosening a physiological security mechanism against exaggerated immune reaction. The target is the antigen 4 on cytotoxic T-Lymphocytes (CTLA-4). CTLA-4 is being installed in the cell membrane after the activation of the immune cell and interrupts signals, which are essential for the clonal expansion and differentiation of T-cells. This break mechanism is normally very sensible, because it prevents, the immune reaction from getting out of control. But when there is tumor cells spreading in the body, a more intensive immune reaction could be useful – so at this point, Ipilimumab plays its role. The antibody binds specifically to CTLA-4 and weakens the break signal. That way Ipilimumab is supposed to unleash the body’s own immune resources for the battle against cancer.
Vemurafenib works completely different. It blocks a mutated protein, which is responsible for the development of the cancer in some of the patients – the BRAF-V600E-kinase. The wild type of this enzyme transduces growth signals from the surface of the cell to the cell core and induces cell proliferation. The mutated form V600E doesn’t require any growth signals to activate proliferation. It activates the signal cascade permanently and thereby leads to malignant degeneration. Vemurafenib blocks the permanent activation through that mutated BRAF-Kinase. For that reason, the kinase-inhibitor Vemurafenib only works for patients who carry that mutated protein – which is present in approximately every second patient.
No light without shadow
The disadvantages of the therapies are connected closely to how the drug works. There is a high risk of immune provoked side effects under Ipilimumab, which usually manifests in the gastrointestinal tract and on the skin.
In its current publication the “Institute for Quality and Profitability in Health Care” reports about a “considerable damage potential” regarding its evaluation of benefit. In total however, the institute attests a substantial additional benefit for the therapy in comparison to “best supportive care”. In the meantime, the institute has also released an evaluation of benefit for Vemurafenib. In it, the institute rates the additional benefit of the drug compared to the comparison therapy as substantial as well.
Regarding the security profile of Vemurafenib, a rising number of squamous epithelium cancer of the skin is apparent. It is suspected, that the inhibition of the BRAF-Kinase in this cases, leads to a paradox activation of the downstream MAPK-section within the signal path. This effect is also being connected to the occurrence of resistances against Vemurafenib. In conclusion, the benefits of the two new therapies are facing serious limitations.
Prof. Dr. Eckhart Kämpgen, leading dermatologist at the Center for Skin Cancer at the University of Erlangen, was asked on his views of the chances and limitations of the two new therapies against malign melanoma. Each week over 100 patients are treated at the centre. His response was that,
“Vemurafenib only works in patients, who carry the BRAF-V600E mutation, but if they do, it works with a high likeliness and very quick. Often, the tumors begin to shrink within a few days, the affected experience this like a miracle. Unfortunately, this effect only lasts for a few months in most cases. Patients, who benefit from it for more than 2 years, is rare. With Ipilimumab the miracle is, that in the studies, some people were alive even after 4 years. Most likely the immune therapy is the only possibility at the moment, to actually heal a patient, because only the immune system is capable of finding single tumor cells in the body and destroying them. A disadvantage of the therapy is, that there has to be immunological resources in the first place, which can be stimulated. This is not the case in patients with a high tumor burden. In addition, Ipilimumab stimulates completely unspecific immune responses. This explains the very high rate of immune related side effects.”
When asked if there is already a kind of therapy-algorhythm that can be taken from the profiles of these drugs? Kämpgen expressed that Vemurafenib is like a high trump in a card game.
“It will win, but you can only use it once. We see its area of use therefore in patients with a very high tumor burden, who need quick help. Is the tumor burden not too big on the other hand, you should not waste this trump and instead use another option, like Dacarbazin. With Ipilimumab you can seek a long time survival in patients with low or therapeutic reduced tumor burden."
When asked how he envisions the treatment of the advanced malign melanoma in a few years, Kämpgen was optimistic in the use of immunotherapy. He said,
“Here it is important to induce immune responses specifically against the tumor cells. We try to achieve that by, using dendritic cells of the patient, presenting the immune system a kind of profile of the tumor cells. Following this vaccination, you can use an unspecific therapy like with Ipilimumab to boost the immune response. We also expect progress from the combination of mutation specific inhibition agents, like Vemurafenib with a MAPK-Inhibitor. This can be more effective than a monotherapy and – as it has shown already –also be also more tolerable.”
Meanwhile researchers at the University of Cologne hospital could show that the combination of the Immunotherapy Ipilimumab with a local therapy can double the survival time of patients with a metastasised Melanoma -compared to a single medication treatment.
The results of the study was published in the renown “Cancer Immunology Research Journal” of the American Association for Cancer Research.
The authors of the study are members of the Cologne workgroup for radio immunooncology (RIO) at the Center for Integrated Oncology Cologne Bonn (CIO) at the University Clinic in Cologne, They include Priv.-Doz. Dr. Sebastian Theurich, Priv.-Doz. Max Schlaak, Dr. Christian Baues and Prof. Dr. Dr. Michael von Bergwelt-Baildon, together with Swiss physicians have studied 127 Melanoma patients, who where treated with Ipilimumab at four centers in Germany and Switzerland.
“The malign melanoma is a very aggressive form of skin cancer, but the Immunotherapy with Iocal therapy has revolutionized the treatment. About 20 percent of the patient, who are only treated with Iocal therapy permamently respond to the therapy. Even though this is already a progress compared to the traditional treatments, we are looking for possibilities, to help more Melanoma patients with this treatment.”, says Priv.-Doz. Dr. Sebastian Theurich.
The trial shows that compared to patients, who only were treated Immunotherapyl with Iocal therapy, those who were treated additionally with radiation therapy and a local therapy, showed a significant prolonged survival time. Local therapies are used for a long time already in the palliative treatment of malign melanoma.
"If you add the local therapy to the immunotherapy, the median survival time of the treated patients doubles from 42 weeks to 93 weeks, without showing more side effects," explains Dr. Max Schlaak, Assistant Director of the Skin Tumor Center of the University Clinic of Cologne. "The local therapies seem to additionally activate the immune response against the tumor cells and are therefore capable to also attack the tumor in other parts of the body", adds Priv.-Doz. Theurich. For the treatment of patients with malign melanoma, the combination of these therapies means new possibilities.
Whether the combination of the immunotherapy and the local therapy can be a treatment option for all cancer patients, this will be further investigated by the Cologne work group RIO. The workgroup RIO (AG RIO) resulted out of the work on this trial in 2016 and investigates all the cancer patients, who are treated at the Center for Integrated Oncology with immunotherapy and local therapy. The members of the AG RIO come from the departments of preclinical research, oncology, dermatology, radiotherapy and radiology.
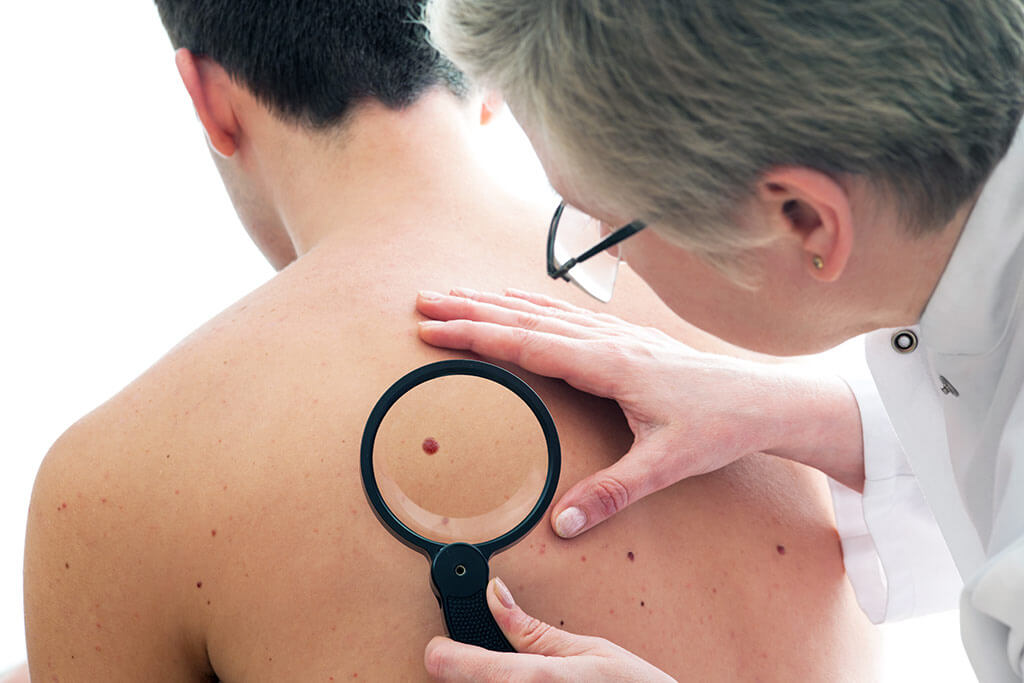
Across the globe in Chicago, Bristol-Myers Squibb presented the results of his new immunotherapy drug at the annual meeting of the American Society of Clinical Oncology. The competitor Merck followed suit. The company also had a drug tested, which effectively fights the tumour by loosening the breaks of the immune system. The effects were dramatic, but the benefit is still questionable.
Since chemotherapies have little effect on melanoma, many drugs have since been tested on this malign tumour, that are supposed to strengthen the immune system in order to fight the tumor.
Interferones are one of the standard therapy for metastasized melanoma, which activate natural killer cells and T-Lymphocytes, and also Aldesleukin (Interleukin 2), a growth factor for T-cells.
The newest development are drugs, which loosen build-in breaks of the immune system. The first agent of that kind is Ipilimumab, which was introduced to Europe in 2011. Ipilimumab blocks the immunomodulating receptor CTLA-4 (Cytotoxic T-Lymphocyte Antigen-4) and unleashes a fierce, but only shortly effective attack of the immune system. In clinical studies Ipilimumab impressed doctors by its fast and sometimes complete remission. But in the end, in this phase-III-study, it only resulted in an average – 4 months extended survival time, which had to be bought with not insignificant side effects. The sense of this therapy is therefore controversial.
The results of a phase-I- trial are now published in the New England Journal of Medicine.
The study involved 88 patients with advanced melanoma, who underwent therapy using Ipilimumab. In the course of the study, the patients received the CTLA-4-blocker Ipilimumab and a second drug, Nivolumab, which blocks the receptor PD-1. CTLA-4 and PD-1 play a role in the modulation of the immune response at different points. The expectation is, that the effects of both drugs add up.
The results were presented by Jedd Wolchok from the Memorial Sloan-Kettering Cancer Center in New York - could not prove to be exact, because there was no real control group. The trial compared a combined therapy, in which both drugs were used simultaneously, with a sequential therapy, in which the patients were further treated with Nivolumab, after Ipilimumab had failed.
Just as in the first study for Ipilimumab, the doctors were amazed by the quick and obvious tumor reduction. In a few patients, the tumors decreased by more than 80%, reported Wolchok in a press release by his clinic, and the remissions lasted more than 12 weeks or longer. But in the study it is then mentioned, that only 5 patients achieved a complete remission, whereby that simply means, that doctors can’t detect any tumors via imaging methods, which doesn’t mean that there are no tumor cells left in the body.
Because of the lack of a control group it is not clear, what the advantages are for the patients. The actual purpose of the phase-1b-study is a safety evaluation and the search for a tolerable dosage. The efficiency is only being tested in later trials. Under the combination, almost all the patients experienced side effects. The most common were skin breaking out, itching, fatigue and diarrhea. Also an increase in lipase and liver enzymes was detected.
More than half of the side effects was determined as grave (grade 3 or 4). At least there was no case of death. For the evaluation of efficiency the dosage they determined was 1 mg/kg body weight for Nivolumab and 3mg/kg body weight for Ipilimumab.
Also the antibody Lambrolizumab, produced by the German company Merck, blocks the PD-1-receptor. The agent, like Nivolumab – is at the beginning of the clinical evaluation. Under the direction of Antoni Ribas of the University of California in L.A., 135 patients with advanced melanoma were treated in a Phase-I-study with different dosages of Lambrolizumab.
Because Lambrolizumab wasn’t combined with Ipilimumab, the tolerance was a little bit better, but there was also a smaller number of complete remissions (6 out of 135 according to table 3), as reported by the publication in the New England Journal of Medicine.
But that doesn’t hold Ribas back from reporting the “highest rate of persisting remissions” of melanoma that he had ever experienced in his practice. The median progression-free survival was stated with 7 months. Here as well there can’t be any statements made regarding the efficiency, because of the lack of a control group, even if the fast progression of the melanoma in the advanced stage gives reason for hope.
Also the participating companies are hopeful. The annual treatment costs with monoclonal antibodies are around 100.000 Euros and both companies are testing if the agents can also be used for other tumours.
Choose treatment abroad and you will for sure get the best results!
Authors:
This article was edited by medical experts, board-certified doctors Dr. Nadezhda Ivanisova, and Dr. Bohdan Mykhalniuk. For the treatment of the conditions referred to in the article, you must consult a doctor; the information in the article is not intended for self-medication!
Our editorial policy, which details our commitment to accuracy and transparency, is available here. Click this link to review our policies.
Sources:
European Society for Medical Oncology
Read:
Immunotherapy for melanoma in Germany
Cancer Treatment - Oncology Clinics in Germany
New Effective Treatments for Stage 4 Cancer: Innovations in Oncology